Expanding the knowledge on the potential of cancer vaccines

Lung cancer is, to this day, the leading cause of cancer-associated deaths worldwide. There is a dire need for the development of more effective therapies, as lung tumors often become resistant to both conventional and targeted therapies, such as immunotherapies. This collaboration will work on advancing a promising cancer therapy referred to as dendritic cell (DC) vaccines.
DCs play a fundamental role in orchestrating the functions of our immune system. They present antigens on their surface that are recognized by other players in the immune response. Their biological role has long been exploited to develop DC vaccines for patients with cancer. The goal of a DC vaccine is to make the patient’s own immune system recognize and eliminate the cancer cells. More specifically, immature DCs are isolated from a patient with cancer and then exposed to tumor-associated antigens. After reaching full maturity, the cells are reintroduced in the patient to trigger an anti-tumor response. However, this traditional approach has several limitations and has yielded mixed clinical results.
This TANDEM project aims to advance the design of DC vaccines for lung cancer therapy. It exploits a novel type of DC, which is engineered in the laboratory to improve its ability to present tumor antigens to the immune system. This work is poised to improve the therapeutic potential of DC vaccines and will hopefully provide a new treatment strategy for lung cancer patients.
Employing new visualization technologies will further the understanding of CAR-T cell therapy

The immune system plays a crucial role in inhibiting tumor growth. Adoptive cell therapy is a kind of immune therapy, where the cells of the patient’s own immune system are extracted, reprogrammed separately, and introduced back into the body to combat tumors in a very targeted manner.
More specifically, an adoptive cell therapy that uses chimeric antigen receptor T cells (CAR-T) has been transformative for people suffering from selected hematological malignancies (cancers that begin in the blood forming tissue) that are prone to relapse or are refractory. CAR-T therapies are being expanded to treat solid tumors as well. Unfortunately, there are significant toxicities associated with CAR-T cell therapy, especially since preclinical tools to evaluate CAR efficiency and safety can be inaccurate, time-consuming, and expensive.
The aim of this TANDEM project is to increase the precision and safety of engineered T cells by implementing new microscopy technologies to dissect and examine the interaction between the CAR-T cell and the tumor cell. They aim to make these technologies easy to implement so that they can routinely be used in a clinical setting. They will in turn provide insight on whether the therapy will be effective or generate toxicity. If successful, this new imaging tool will be used to improve both the patient’s prognosis and the patient’s overall quality of life during CAR-T treatment of hematological malignancies.
Understanding disease progression in lung cancer

Lung adenocarcinoma (LUAD) represents 40% of all lung cancers, which makes it one of the most common lung cancers. Previous studies have used histopathology (the study of changes in tissues caused by disease) to diagnose and study diseased lung tissue at the microscopic level. This classic approach identified changes in cell morphology and growth patterns that accompany disease progression. The overarching aim of the current collaboration is to use new techniques to predict more accurately how the cancer will progress and whether or not it will react to treatment.
Disease progression is driven by the plasticity of cell identity and a coincident reshaping of the tumor environment, such that reprogramming is maintained. Based on histopathological analysis, there are four recurrent tumor progression patterns that reflect both tumor aggressivity and patient survival prognosis. The milestones are readily identified and provide a lot of information on disease progression and tumor heterogeneity, both within one patient and among multiple patients:
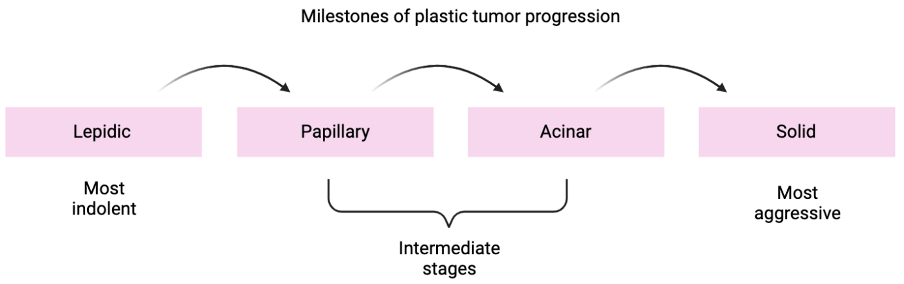
Whereas it is clinically relevant to identify the stages of tumor progression by histopathology, the molecular drivers of the transitions from one state to the next are also crucial. Towards this end, the team has characterized molecularly the transitions of cancer from lepidic to solid tumors, using a combination of techniques for single cell analysis of cancer cells and their interaction with the tumor microenvironment.
The team has the following aims: first is to detect molecular features of the transitions from lepidic to solid tumors across patients (see figure). Secondly, they will deepen our understanding of how tumor progression can be predicted based on the interaction of cancer cells with the tumor cell environment. The overarching goal of the project is to provide new insights into the role of cancer cell plasticity in disease progression and to explore if this helps predict disease progression in individual patients. Ultimately, the results will pave the way for new practices in clinical diagnostics as well as new approaches to lung cancer therapy.
TCR-engineered CD4 T cell transfer for optimized cancer immunotherapy in adult and pediatric patients

This is a highly translational project aiming at harnessing the tumoricidal activities of CD4+ T cells to optimize cancer immunotherapies. The project includes preclinical validation of TCR-engineered CD4 T cells and the setup of a Phase 1 clinical trial for relapsed and refractory solid tumors in both adult and childhood cohorts.

